THERAPEUTIC
PLASMA EXCHANGE
for Alzheimer’s Disease

A New Frontier in Alzheimer’s Treatment
Clinical Outcomes
Slowed Cognitive Decline
Multiple studies demonstrate that TPE-treated patients experience significantly less cognitive deterioration than controls, with benefits observed in memory, language, processing speed, and executive function — particularly in moderate-stage disease.
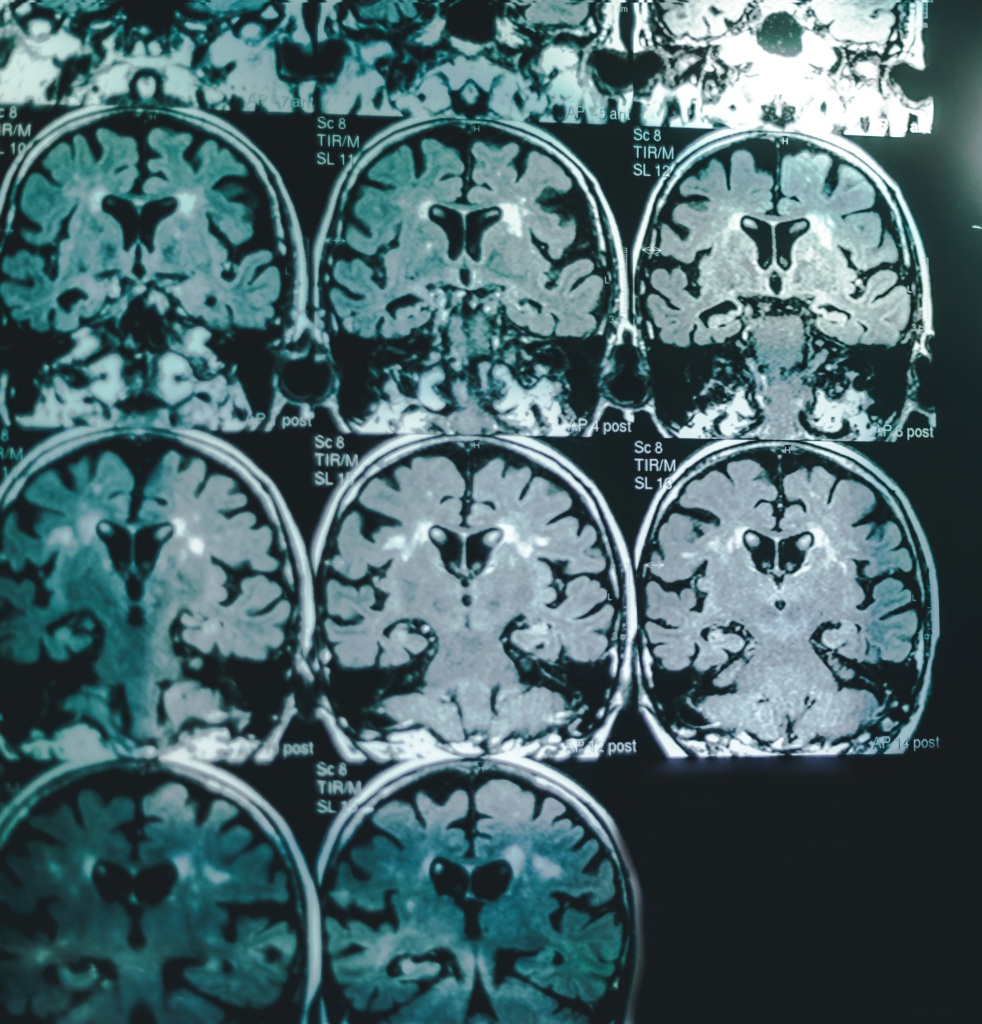
Brain Structure Preservation
Neuroimaging data from the AMBAR program show better preservation of hippocampal volume, reduced subcortical atrophy, and less metabolic decline on FDG-PET in treated patients, suggesting a protective structural effect on the brain.
Neuropsychiatric Improvement
TPE-treated patients show reductions in behavioral and neuropsychiatric symptoms including agitation, depression, and anxiety. These improvements contribute to enhanced quality of life for both patients and caregivers.
Overview of Therapeutic Plasma Exchange for Alzheimer’s
Alzheimer’s disease is the most common cause of dementia, accounting for approximately 70% of cases worldwide. It is characterized by the progressive accumulation of amyloid-beta (Aβ) plaques and neurofibrillary tangles of phosphorylated tau protein, leading to neuronal damage, chronic neuroinflammation, and the gradual loss of memory, reasoning, and functional independence.
Despite decades of research, curative options remain unavailable, and most current treatments focus on symptomatic relief rather than disease modification. However, therapeutic plasma exchange (TPE) with albumin replacement has emerged as a promising intervention that may alter the underlying disease trajectory — supported by the landmark AMBAR clinical trial and growing real-world clinical evidence.
TPE represents a fundamentally different approach: rather than targeting amyloid in the brain directly, it leverages the body’s own equilibrium between central and peripheral Aβ to encourage clearance from the central nervous system while simultaneously replenishing neuroprotective albumin.
The Scientific Rationale
The scientific foundation for Therapeutic Plasma Exchange for Alzheimer’s disease rests on the peripheral sink hypothesis — the principle that amyloid-beta in the cerebrospinal fluid exists in a dynamic equilibrium with Aβ circulating in the blood. Approximately 90% of plasma Aβ is bound to circulating albumin. By removing Aβ-laden plasma through therapeutic exchange and replacing it with fresh albumin, TPE creates a concentration gradient that encourages Aβ to exit the brain across the blood-brain barrier.
This concept has been validated through multiple lines of evidence. In preclinical studies, peripheral reduction of Aβ levels has been shown to decrease brain amyloid burden. In clinical studies, TPE has produced measurable changes in both plasma and cerebrospinal fluid amyloid concentrations, with a characteristic “sawtooth” pattern observed: Aβ levels drop after each exchange and return toward baseline before the next procedure, confirming the dynamic equilibrium.
Beyond amyloid clearance, TPE offers additional therapeutic mechanisms. The albumin found in Alzheimer’s patients is often oxidized and glycated, reducing its protective capacity. Replacing degraded albumin with functional therapeutic albumin restores antioxidant capacity and the ability to bind and neutralize Aβ. Furthermore, TPE removes circulating inflammatory mediators — cytokines and chemokines — that contribute to the neuroinflammation increasingly recognized as a driver of disease progression.
A Look at the Research
Therapeutic Plasma Exchange for Alzheimer’s Disease
Alzheimer’s Management By Albumin Replacement (AMBAR)
The AMBAR trial is the largest and most comprehensive investigation of TPE for Alzheimer’s disease to date. This multicenter, randomized, blinded, placebo-controlled phase 2b/3 trial enrolled 347 patients with mild-to-moderate AD across 41 clinical sites in Spain and the United States. Patients were randomized to one of three active treatment groups receiving plasma exchange with varying doses of albumin and IVIG, or a sham (placebo) group.
The treatment protocol consisted of a 6-week intensive phase with weekly conventional plasma exchange, followed by a 12-month maintenance phase with monthly low-volume plasma exchange. The study measured changes in cognitive function (ADAS-Cog), daily functioning (ADCS-ADL), clinical dementia staging, cerebrospinal fluid biomarkers, and brain structure and metabolism through neuroimaging.
Real-World Study: TPE Safety & Efficacy in Alzheimer’s Patients
Taragano F, Seinhart D, Epstein P, et al. — Journal of Alzheimer’s Disease, 2025
This prospective cohort study evaluated 32 patients with mild-to-moderate AD treated with TPE in a clinical setting in Argentina between 2022 and 2024, compared against 194 historical controls. Over 514 total procedures, TPE-treated patients showed 45% less decline on MMSE scores, 88% less deterioration in immediate recall, and 74% less decline in delayed recall compared to controls. Language and executive function were also better preserved. No severe adverse events were reported, with over 81% of sessions uneventful. This study provides the first independent real-world validation of the AMBAR findings.
Scoping Review: Plasma Exchange with Albumin in AD Management
Cantero-Fortiz Y, Boada M — Frontiers in Neurology, 2024
This comprehensive scoping review evaluated seven studies from the AMBAR trial program, finding consistent evidence across all studies for improvements in cognitive function, positive neuroimaging outcomes, and favorable neuropsychiatric results. The review confirmed that TPE-treated patients showed 15% better hippocampal volume preservation and 10% improvement in brain perfusion metrics compared to controls. The safety and tolerability profile was consistently favorable across all evaluated studies.
A Note on the Evolving Clinical Landscape
TPE with albumin replacement occupies a unique position in the Alzheimer’s treatment landscape. Unlike cholinesterase inhibitors and memantine, which address symptoms, and unlike anti-amyloid monoclonal antibodies (such as lecanemab and donanemab), which target brain amyloid directly, TPE works through peripheral amyloid clearance combined with albumin replenishment — a distinct mechanism not shared by any other currently available intervention.
While TPE remains an emerging approach and is not yet approved as a standard-of-care therapy for Alzheimer’s disease, the clinical evidence base is growing. The AMBAR trial demonstrated meaningful disease-slowing effects in a rigorous placebo-controlled setting, and these findings have now been independently supported by real-world data. Ongoing research continues to refine the optimal dosing regimen, patient selection criteria, and combination strategies.
Our practice offers TPE as part of a comprehensive, individualized approach to Alzheimer’s care, integrating the best available evidence with expert clinical judgment and close collaboration with patients and their families.
Additional References
Boada M, López OL, Olazarán J, et al. A randomized, controlled clinical trial of plasma exchange with albumin replacement for Alzheimer’s disease: Primary results of the AMBAR Study. Alzheimers Dement. 2020;16(10):1412–1425.
Taragano F, Seinhart D, Epstein P, et al. A real-world study on the safety and efficacy of therapeutic plasma exchange in patients with Alzheimer’s disease. J Alzheimers Dis. 2025;108(1):129–141.
Cantero-Fortiz Y, Boada M. The use of plasma exchange with albumin replacement in the management of Alzheimer’s disease: a scoping review. Front Neurol. 2024;15:1443132.
Cuberas-Borrós G, Roca I, Castell-Conesa J, et al. Neuroimaging analyses from a randomized, controlled study to evaluate plasma exchange with albumin replacement in mild-to-moderate Alzheimer’s disease: additional results from the AMBAR study. Eur J Nucl Med Mol Imaging. 2022;49(13):4589–4600.
Gonzalo R, Minguet C, Ortiz AM, et al. Plasma exchange with albumin replacement for Alzheimer’s disease treatment induced changes in serum and cerebrospinal fluid inflammatory mediator levels. Ann Clin Transl Neurol. 2024;11(12):3280–3291.
Rohrer L, Yunce M, Montine TJ, Shan H. Plasma Exchange in Alzheimer’s Disease. Transfus Med Rev. 2023;37(1):10–15.
Imbimbo BP, Ippati S, Ceravolo F, Watling M. Perspective: Is therapeutic plasma exchange a viable option for treating Alzheimer’s disease? Alzheimers Dement Transl Res Clin Interv. 2020;6(1):e12004.
Boada M, Anaya F, Ortiz P, et al. Efficacy and safety of plasma exchange with 5% albumin to modify cerebrospinal fluid and plasma amyloid-β concentrations and cognition outcomes in Alzheimer’s disease patients: a multicenter, randomized, controlled clinical trial. J Alzheimers Dis. 2017;56:129–143.
